(Aminomethylene)bisphosphonic acid (AMBP) is widely used as a scale inhibitor and metal ion chelating agent in industrial water treatment systems. Despite its frequent application in cooling towers, boilers, and high-hardness water environments, confusion sometimes arises regarding its chemical classification. Specifically, is (Aminomethylene)bisphosphonic acid truly a phosphonate, or does it belong to another phosphorus-containing category?
From a structural and chemical standpoint, the answer is clear: AMBP is an organophosphonate, and more precisely, it is an aminobisphosphonate. Understanding why requires a closer look at the definition of phosphonates, the molecular structure of AMBP, and the differences between phosphonates and phosphates.
Understanding the Definition of a Phosphonate
A phosphonate is an organophosphorus compound characterized by the presence of a direct carbon–phosphorus (P–C) bond. This bond is the defining feature that distinguishes phosphonates from inorganic phosphates. In phosphates, phosphorus atoms are connected to oxygen atoms through P–O bonds, whereas in phosphonates, at least one phosphorus atom is directly bonded to carbon.
This structural distinction is not merely academic. The P–C bond provides greater hydrolytic stability, meaning phosphonates are far more resistant to breakdown under high temperature or alkaline conditions. Because of this stability, organophosphonates are extensively used in industrial water treatment chemicals, especially in applications requiring reliable long-term scale inhibition.
Phosphonates are often referred to as phosphonic acids or their salts, and they function primarily as chelating agents and threshold scale inhibitors. If a compound contains the P–C linkage and behaves chemically as a phosphonic acid derivative, it qualifies as a phosphonate. AMBP meets these criteria unequivocally.
Chemical Structure of (Aminomethylene)bisphosphonic Acid
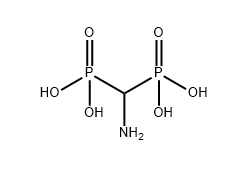
The molecular structure of (Aminomethylene)bisphosphonic acid contains two phosphonic acid groups attached to the same central carbon atom, along with an amino functional group. This arrangement forms what is known as a P–C–P backbone. The presence of two phosphonic acid groups confirms that the compound contains direct carbon–phosphorus bonds, thereby satisfying the structural definition of a phosphonate.
The amino group attached to the central carbon further classifies the molecule as an aminophosphonate. Structurally, AMBP can be described as an aminomethylene derivative in which the methylene carbon serves as the anchor point for both phosphonate groups.
This P–C–P configuration is responsible for the compound’s strong chelation capacity. The phosphonic acid groups can coordinate tightly with metal ions such as calcium, magnesium, and iron. In industrial water systems, this coordination ability enables AMBP to interfere with crystal growth and prevent scale formation. The structure therefore determines both its classification and its practical function as a high-efficiency scale inhibitor.
What Makes a Compound a Bisphosphonate?
The term “bisphosphonate” refers to a specific subclass of phosphonates in which two phosphonate groups are attached to the same carbon atom. The general structural formula can be represented as R–C(PO₃H₂)₂, where the substituent R may vary. In the case of (Aminomethylene)bisphosphonic acid, the R group includes an amino functionality.
Because AMBP contains exactly two phosphonic acid groups bonded to one carbon center, it fits squarely within the bisphosphonate category. More specifically, it is classified as an aminobisphosphonate due to the presence of the amino substituent.
This classification is important in both chemical theory and industrial practice. Bisphosphonates are typically stronger chelating agents than monophosphonates because they provide multiple coordination sites for metal ions. In water treatment formulations, this structural advantage translates into improved calcium tolerance and enhanced threshold inhibition performance. As a result, AMBP is often selected for systems operating under high hardness or high temperature conditions.
For comparison, other widely used industrial phosphonates include HEDP and ATMP. While each compound differs in functional groups and performance profile, they all share the defining P–C bond that places them in the phosphonate family.
Phosphonate vs Phosphate: Key Differences
One of the most common misunderstandings in industrial chemistry is the assumption that phosphonates and phosphates are interchangeable. Although both contain phosphorus, their chemical structures and performance characteristics differ significantly.
Phosphates are inorganic compounds characterized by phosphorus–oxygen linkages. They may form polyphosphate chains through P–O–P bonds, but they do not contain direct carbon–phosphorus bonds. As a result, phosphates are generally more susceptible to hydrolysis and thermal degradation, especially under high-temperature boiler conditions.
Phosphonates, by contrast, possess at least one direct P–C bond. This bond is significantly more resistant to chemical breakdown. The enhanced stability allows phosphonates to maintain their scale inhibition properties in demanding environments such as cooling towers and recirculating water systems.
Functionally, phosphonates like AMBP act primarily through threshold inhibition, crystal distortion, and metal ion sequestration. Traditional phosphates may function as precipitation inhibitors but often require higher dosages and may contribute to higher phosphorus discharge levels. From both a performance and regulatory standpoint, distinguishing between phosphonate and phosphate chemistry is essential.
Why Correct Classification Matters in Industrial Applications
Accurately identifying (Aminomethylene)bisphosphonic acid as a phosphonate has practical implications beyond chemical nomenclature. In regulatory documentation, safety data sheets, and product labeling, precise classification ensures compliance with chemical reporting standards. Mislabeling a phosphonate as a phosphate could lead to incorrect environmental reporting or misunderstandings regarding treatment chemistry.
For formulators and engineers, recognizing AMBP as an aminobisphosphonate helps determine its suitability in blended water treatment programs. Its high chelation strength, thermal stability, and compatibility with polymer dispersants and corrosion inhibitors make it a strategic component in advanced industrial formulations.
Procurement professionals also benefit from clear classification. When technical specifications call for a phosphonate-based scale inhibitor, AMBP meets the structural and functional criteria required for such applications. Understanding its chemical identity supports better purchasing decisions and more accurate technical communication between suppliers and end users.



